AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Hydrogen atom12/20/2023  Hydrogen is an energy carrierĮnergy carriers allow the transport of energy in a usable form from one place to another. Hydrogen combined with carbon forms different compounds-or hydrocarbons-found in natural gas, coal, and petroleum. Hydrogen combined with oxygen is water (H2O). Hydrogen occurs naturally on earth only in compound form with other elements in liquids, gases, or solids. The sun is essentially a giant ball of hydrogen and helium gases. Stars such as the sun consist mostly of hydrogen. Hydrogen is also the most abundant element in the universe. Each atom of hydrogen has only one proton. Hydrogen is a gas at normal temperature and pressure, but hydrogen condenses to a liquid at minus 423 degrees Fahrenheit (minus 253 degrees Celsius). “You can think about our experiment as a tool that allows you to look inside the atom and see what’s going on,” Stodolna says.Energy Kids: U.S. Crucially, the pattern was a projection of the spacings of the energy levels in the hydrogen atom, as laid out in the wave function, with bright rings where electrons were present and dark lanes where they were not ( Physical Review Letters, doi.org/mmz). The electrons emitted waves that produced an interference pattern on the detector (see “An atom undressed”). An applied electric field then pushed the excited electrons away from their respective nuclei, towards a detector about half a metre away. Stodolna’s team made a beam of atomic hydrogen and zapped it with two separate lasers that excited the atoms’ electrons by precise amounts. Do it with enough atoms and the number choosing each spot will reflect the quantum probabilities laid out by the wave function. 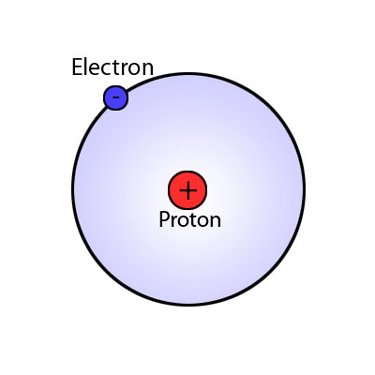
This removes the quantum nature of each individual atom’s electron, forcing it to choose a particular location from those it is allowed to reside in. Rather than taking an image of a single atom, they sampled a bunch of atoms. Her team decided to make a picture using a technique dreamed up 30 years ago that can be thought of as a quantum microscope. They’re exquisite quantum objects that change their appearance upon observation,” says Aneta Stodolna of the FOM Institute AMOLF in Amsterdam, the Netherlands. “Wave functions are difficult to measure. In principle, the wave function, denoted by Greek letter psi, can be used to reveal these energy levels for any given atom or molecule, although in practice this has only been done for the very simplest – the hydrogen atom and molecule (made of two hydrogen atoms bonded together).īut how on earth do you make an image of such an object? Measuring the position of a single electron “collapses” the wave function, forcing it to pick a particular position, but that alone is not representative of its normal, quantum presence in the atom. 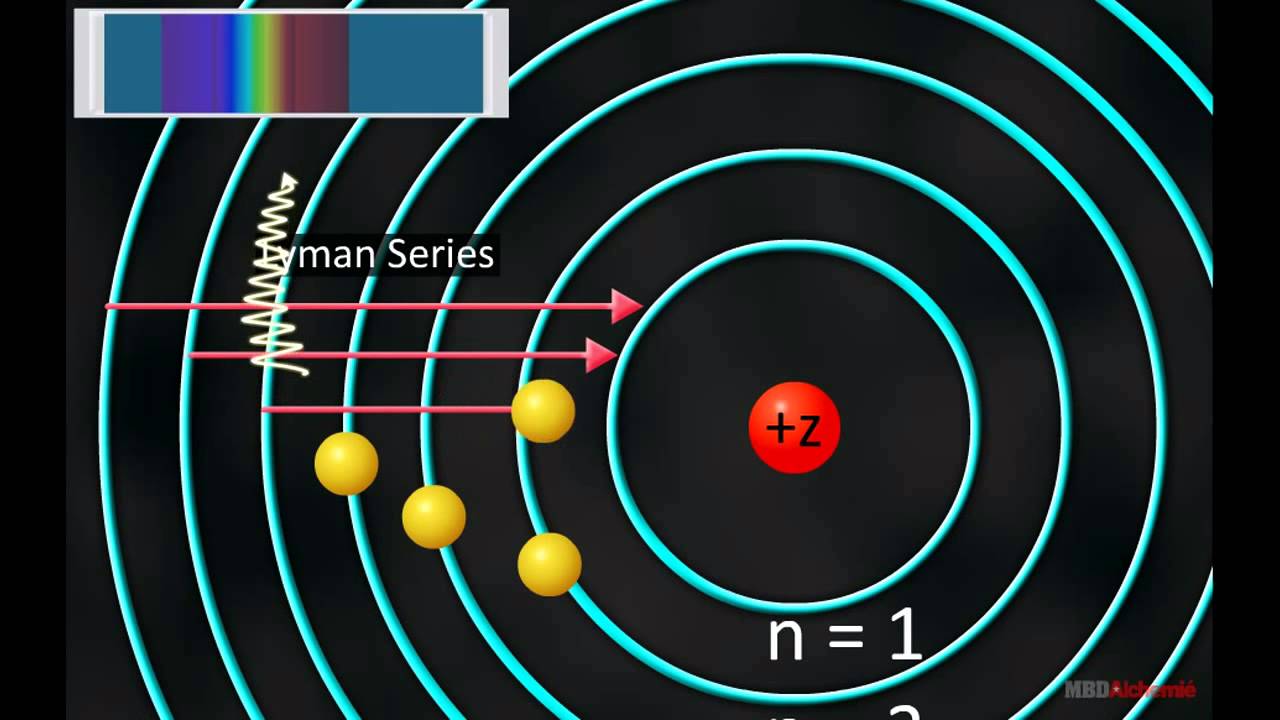
Not all positions are equally available: electrons can only reside at certain distances from the nucleus, with these distances related to how much energy the electron holds. 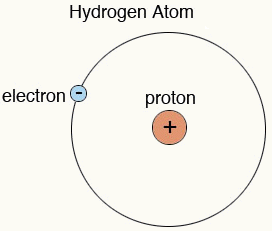
The best you can do is say how likely it is to be in a given spot. Thanks to quantum theory, which says that tiny particles are in multiple places simultaneously, you can never say where a given electron actually is. 
The electrons that dance around an atomic nucleus help determine how the atom bonds with others, but they are notoriously difficult to pin down. A quantum-style microscope has imaged the hydrogen atom’s wave function, the equation that determines its electrons’ positions – and in turn the atom’s properties. AN ATOM’S electrons are an ever-shifting quantum melee, but it turns out you can still take their photograph as if they were standing still. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
